 Ionic compounds are compounds that are made up of two or more ions, which. 
The above differences also help distinguish and identify whether a given unknown compound is ionic or molecular, by conducting appropriate tests in a lab, and finding out the differences in chemical and physical properties of the compound. Chemical bonds are formed when atoms lose, gain, or share valence electrons to completely fill their outermost electron shell. Molecular compounds are molecules that have two or more atoms of different elements. This video attempts to clarify this by showing structural differences. Ionic compounds have a higher melting and boiling point than molecular compounds. Students often misunderstand how a formula unit is different than a molecular formula. As a general rule of thumb, compounds that involve a metal binding with either a non-metal or a semi-metal will display ionic bonding.Molecular compounds are softer and more flexible as compared to ionic compounds.Molecular compounds are more flammable than ionic compounds.The enthalpy of fusion (heat energy absorbed when a solid melts) and enthalpy of vaporization (heat energy absorbed when a liquid boils) are higher in ionic compounds.Covalent compounds usually form from two nonmetals. Compounds that do not contain ions, but instead consist of atoms bonded tightly together in molecules (uncharged groups of atoms that behave as a single unit), are called covalent compounds. Ionic compounds are more reactive than molecular compounds. Ionic compounds generally form from metals and nonmetals. Ionic and molecular (covalent) compounds also look different at the microscopic level: covalent and molecular compounds exist in molecules, while ionic compounds are organized in. 
A molecular compound cannot conduct electricity in any state, whereas an ionic compound, when dissolved in an aqueous solution, can act as a good conductor of electricity. Ionic compounds are charged ions, whereas molecular compounds consist of molecules.Ionic compounds are charged ions, whereas molecular compounds consist of molecules.In ionic compounds, the ions are held together due to electrical attraction, whereas, in molecular compounds, the atoms are held together by the attraction between the atoms due to shared electrons. 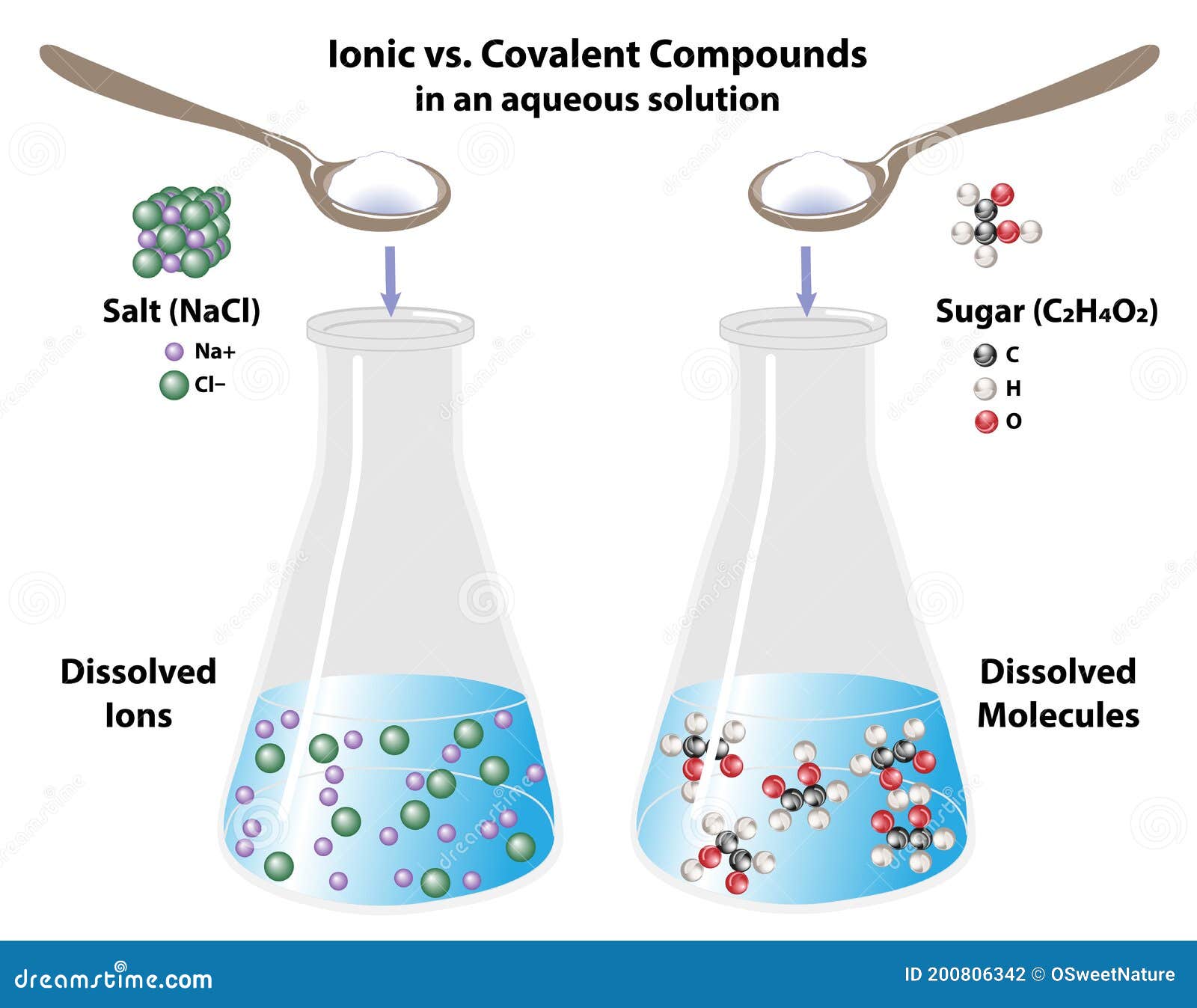
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
